Sialic acid is a terminal cell-surface glycan that modulates multiple immune regulatory pathways. In disease, sialic acid signaling can be co-opted to drive immune dysfunction — enabling tumor cells to evade immune recognition in cancer, and to sustain activation of pro-fibrotic macrophage in autoimmunity.
We are developing engineered sialidase therapies that remove excessive cell-surface sialic acid, restoring normal immune signaling. In cancer, this exposes tumor cells to immune clearance; in autoimmunity, it selectively reduces the pro-fibrotic macrophage populations that drive chronic fibrosis without depleting systemic macrophage populations.
Palleon’s EAGLE (Enzyme-Antibody GLycan Editing) platform fuses human sialidase enzymes with antibodies, enabling tissue- and target-selective sialic acid removal. The platform supports therapeutic applications in both autoimmunity and cancer.
A New Frontier in Biology
Palleon Scientific Co-Founder and Nobel laureate Carolyn Bertozzi’s discoveries in the field of glycobiology enabled the development of Palleon’s therapeutic platform.

Macrophages in Autoimmunity
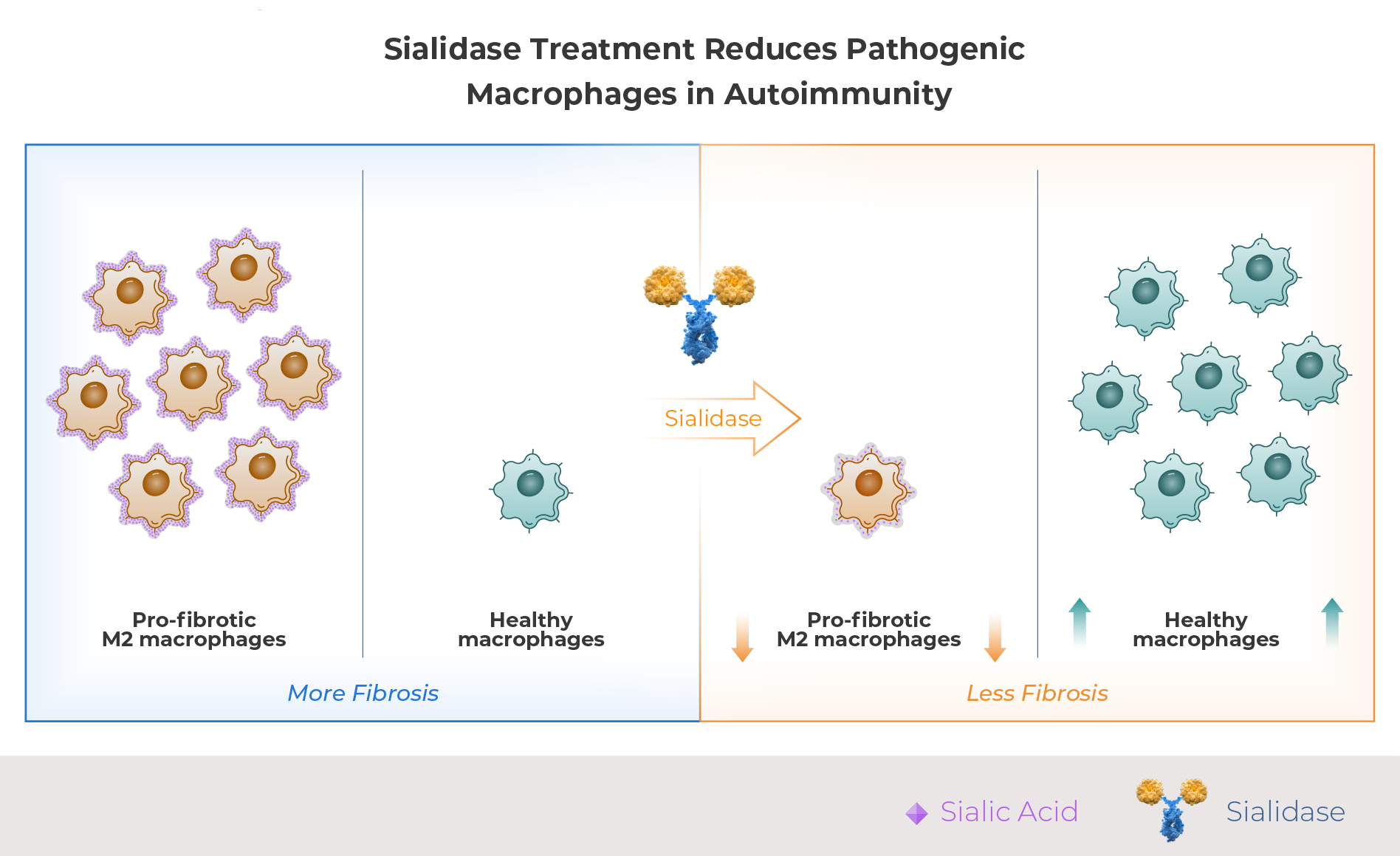
In healthy tissue, macrophages clear debris, resolve inflammation, and orchestrate repair. In autoimmune disease, recurrent inflammatory flares drive chronic macrophage activation — the normal wound-repair program becomes self-perpetuating, progressively replacing functional tissue with fibrotic scar.
CD163⁺ M2 macrophages are a subset found in tissue that drives this process. In normal wound healing CD163+ M2 macrophages are beneficial, but when chronically activated they sustain collagen deposition and suppress inflammatory resolution — converting a protective response into progressive organ fibrosis.
Persistence of these pro-fibrotic macrophages is a documented hallmark of organ damage across a broad range of autoimmune diseases, including membranous nephropathy, lupus nephritis, ANCA vasculitis, IgA nephropathy, RA-associated ILD, systemic sclerosis, and multiple sclerosis. No approved therapy interrupts this fibrotic cycle.
The Sialidase Opportunity in Autoimmunity
Sialic acid is a terminal cell-surface glycan that serves as a key ligand for multiple immune regulatory receptors. Several sialic acid-dependent pathways sustain pathological macrophage activation and impair inflammatory resolution in autoimmune disease.
Palleon’s lead candidate, E-602 (efgitasialase alfa), is a human sialidase-Fc fusion protein that removes sialic acid from cell surfaces, modulating macrophage activation state. E-602 selectively reduces the persistence of pro-fibrotic CD163⁺ M2 macrophages in inflamed tissues without depleting systemic macrophage populations — an important differentiator given the critical role macrophages play in maintaining homeostasis throughout the body.
Clinical Evidence
E-602’s mechanism has been validated in human tissue. In a completed oncology trial with paired biopsies, E-602 reduced CD163⁺ M2 macrophages by more than 50% in 8 of 10 patients — a pre-specified pharmacodynamic endpoint establishing proof-of-mechanism at clinical dose levels. This dataset provided the scientific rationale for advancing E-602 into autoimmunity.
In an ongoing Phase IIa trial in membranous nephropathy, urinary soluble CD163 — a non-invasive, kidney-specific biomarker of local macrophage activity — is being used to confirm target engagement in the kidney.
Targeted Sialidase Molecules in Cancer
Most solid tumors — including lung, ovarian, colon, and prostate cancer — are hypersialylated. Tumor cells upregulate sialyltransferases to dramatically increase sialic acid on their surface engaging inhibitory Siglec receptors on T cells, macrophages, and other innate immune cells. This suppresses immune surveillance and promotes a tumor-associated macrophage phenotype that builds protective stroma.
E-688 combines a human sialidase enzyme with a targeting arm that binds tumor antigen B7-H3. By enzymatically removing sialic acid from tumor cell surfaces, E-688 restores immune recognition, disrupts the immunosuppressive tumor microenvironment, and has potential to generate durable anti-tumor immune responses and memory.
HYDRA Platform
Palleon has developed the powerful HYDRA platform to support our portfolio of therapeutic candidates. HYDRA is an immunohistochemistry-based translational research technology that measures the density of hypersialylation on tissue samples. It provides a clinical biomarker and could potentially be used in the future as a companion diagnostic.