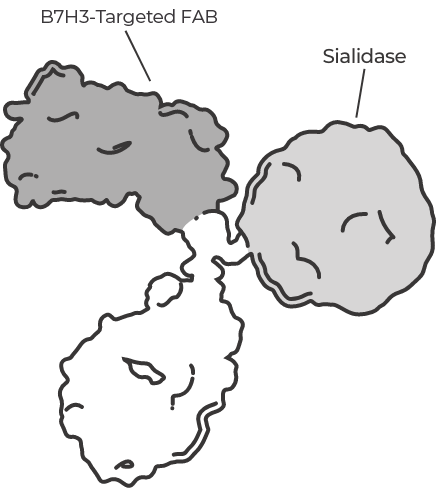
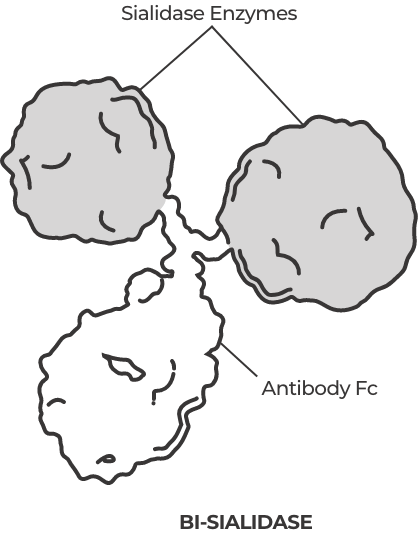
We’re advancing a pipeline of pioneering therapies in oncology and autoimmune diseases, enabled by our groundbreaking scientific discoveries in glycobiology.
Oncology
Discovery
Lead-Op
IND-Enabling
Phase 1
Phase 2
IND-Enabling
Autoimmunity
Discovery
Lead-Op
IND-Enabling
Phase 1
Phase 2
Phase 2
Lead-Op